The first step that needs to be followed while balancing chemical equations is to obtain the complete unbalanced chemical equation. The reactant side is the chemical equation part to the left of the ‘→’ symbol, and the product side is the part to the right of the ‘→’ symbol.ġ. The Inspection approach was used to complete the entire balancing procedure.Įvery chemical equation is a symbolic representation of a chemical reaction where their respective chemical formulae denote the products and reactants.Ĭonsider an example of a chemical equation, 2H 2 + O 2 → 2H 2 O describing the reaction between hydrogen and oxygen to form water Once all atoms and charges are equal, eliminate fractions and find the group of minimal coefficients. You can also use fractions.Ītoms must be balanced at the end of most compounds. Try to balance the atoms in as few compounds as possible. You must begin with the most difficult compound. Let us first go over the technique and then look at an example:Īssume that only one molecule of this chemical is involved in the process. You can determine the proper balance by looking at the equation. Inspection Technique: It refers to the act of inspecting something, and it is used in conjunction with the Inspection method. There are several approaches to balancing a chemical equation, and we'll go over each one. 
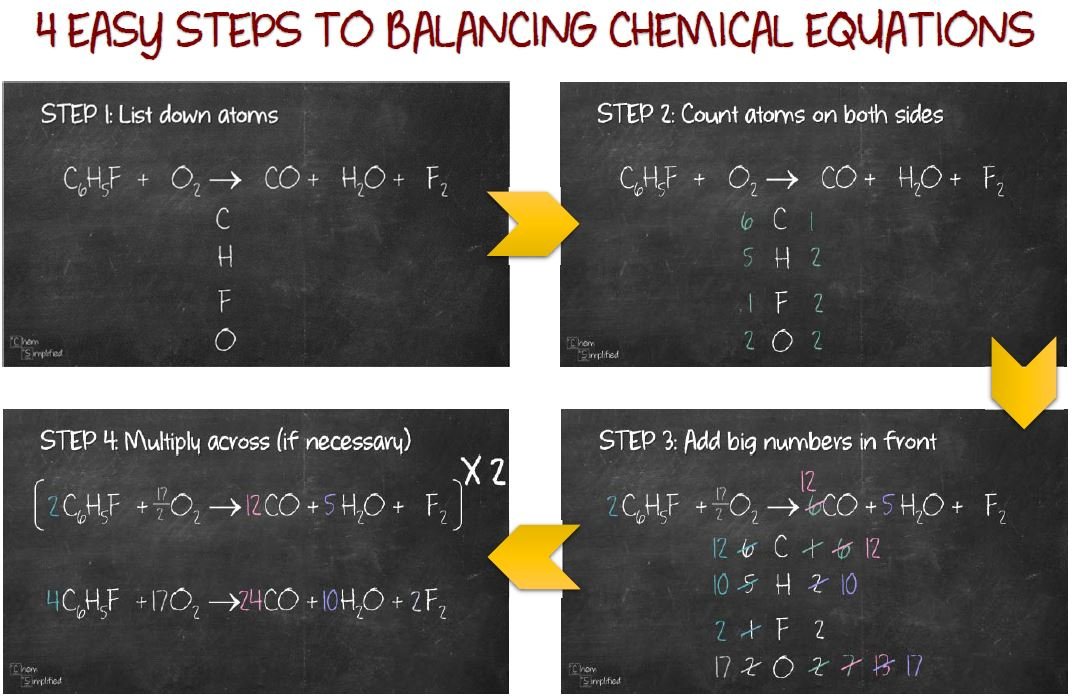
How to Proceed with the Balance of Equations? The hydrogen and oxygen atoms are the easiest to balance last.Ĭheck your work by indicating the state of matter of the reactants and products.Ī few of the things that should be addressed before explaining the two methods are the chemical equation. To make the number of atoms of each element the same on both sides of the equation, multiply coefficients (the numbers in front of the formulas). To show the reactants and products, write the imbalanced equation.Ĭalculate the number of atoms of each element on each side of the reaction arrow. To balance a chemical equation, follow these four simple steps: When the matter is reorganized to generate a different or new material, it is called a chemical reaction.īalancing Chemical Equations - Balancing Chemical Equations Entails Correctly Writing the Chemical Equation so that the Mass on Each Side of the Arrow is Equal Every day, you may see chemical reactions taking place. It's crucial to understand what a chemical reaction is since a chemical equation depicts one. A chemical equation is a technique of representing the components of a chemical reaction in a concise manner. The first one is the traditional balancing equations method and the second method is the algebraic balancing method.Ī chemical equation is a symbolically represented chemical reaction in the form of symbols and formulae, with the reactant entities on the left and the product entities on the right. Let us discuss two easy and faster methods of balancing a chemical equation now. This is necessary because any chemical equation must obey the law of constant proportions and the law of conservation of mass, i.e., the same number of atoms of each element must exist on the product side and reactant side of the equation. Let us discuss balancing chemical equations.īalancing the chemical equations involves the addition of stoichiometric coefficients to products and reactants. The number of atoms of the reactants and products supposed to be balanced. Enter the value 1 in the bottom right cell I8, and fill column K with zeros and 1 as shown.A chemical equation represents the chemical formulas of substances that react and the substances that produce. For the products, column G chromium pentoxide has -5 O and -1 Cr, column H potassium sulphate has -2 K, -1 S and -4 O, and column I water has -2 H and -1 O. Thus in column D, hydrogen peroxide has 2 H and 2 O atoms in column E potassium dichromate has 2 K, 7 O and 2 Cr and in column F sulphuric acid has 2 H, 4 O and 1 S. The table is filled with the numbers of each type of atom in the reactant and product compounds, reactants being positive and products negative. In column J are entered each of the atomic species in the reaction, ie H,K,O,Cr & S ( their order doesn't matter ) and x at the end. Next is a table in block D3:K8 which will contain the input matrix, with the coefficient headings a-f at the top of the block. Column C is for the calculated results and can be left empty to begin with. The reactants and products are entered in column A and their coefficients a,b,c, d,e, & f in column B. The next step is to set up a table in the spreadsheet as shown in Table 1. a H2O2 + b K2Cr2O7 + c H2SO4 → d CrO5 + e K2SO4 + f H2O 
To begin with, we write out the reactant and product species formulae and apply algebraic coefficients to each: Reactants ProductsĮg. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
